UNIT OVERVIEW AND GOALS
1.1 INTRODUCTION TO YEAR 10 SCIENCE: Designing an Experiment
HOMEWORK FOR SECTION 1.1
10GR HomeworkHomework 1.1: Finish all questions in chemistry workbook pages 4-6, including a graph of data collected.
|
10BE HomeworkHomework 1.1: Finish all questions in chemistry workbook pages 4-6, including a graph of data collected.
|
By the end of this section, you should be able to:
- Identify the dependent and independent variables for given experiments and justify your choice.
- Determine possible control variables and explain why they might need to be controlled.
- Write an Experimental Aim (purpose statement) that includes reference to the independent and dependent variables.
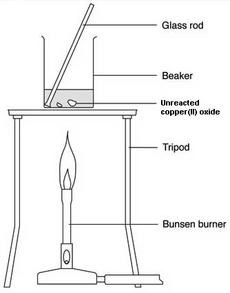
- Write a clear, concise method that includes a properly labeled diagram of the experimental set-up.
- Collect data using a properly labeled data table
- Graph the results on properly labeled graph
- Interpret the results and write a concise conclusion statement
Aim/Purpose
The Aim, or Purpose, Statement is one sentence that describes why you are conducting this experiment or what you hope to find.
Example: "To determine how the concentration of Hydrochloric acid effects the rate of a reaction with Magnesium metal".
Example: "To determine how the concentration of Hydrochloric acid effects the rate of a reaction with Magnesium metal".
VariablesWhen conducting a science experiment, it is important to be able to identify the key variables.
|
|
MethodYour method is the procedure that you carry out during your experiment.
|
Collecting DataYou should have a clearly labeled data table set up before you take your first measurement.
Your data table should include:
|
|
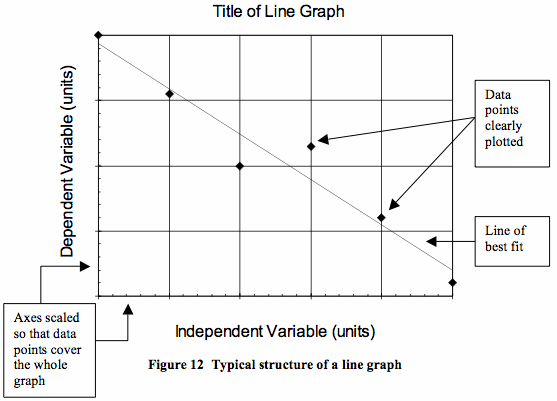
Graphing Your DataA graph is a visual representation of your data and can be used to see trends; how your dependent variable changes as you change the independent variable.
All graphs need:
|
Conclusion
The conclusion is a one or two sentence statement that summarizes your findings based on your data and graph.
Example: We found that the reaction between Hydrochloric Acid and Magnesium was faster with higher concentrations of acid.
Example: We found that the reaction between Hydrochloric Acid and Magnesium was faster with higher concentrations of acid.
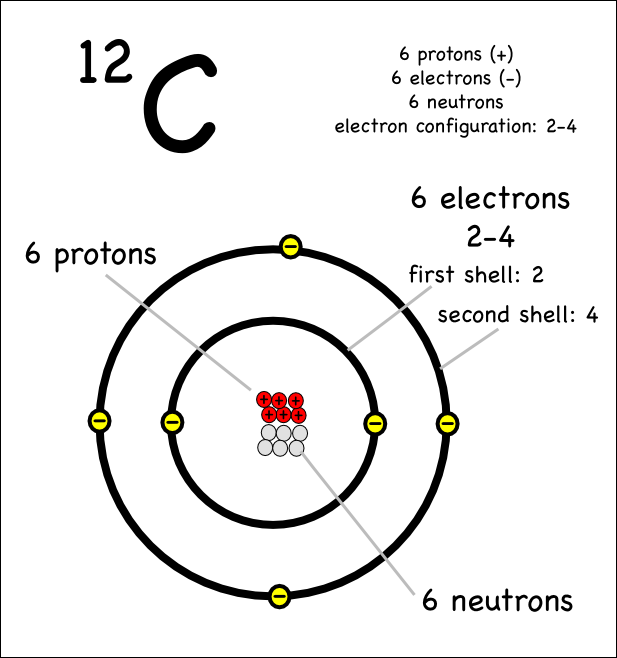
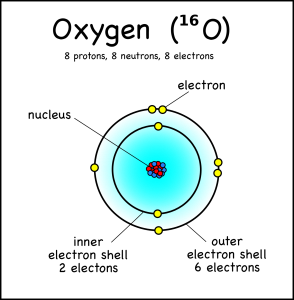
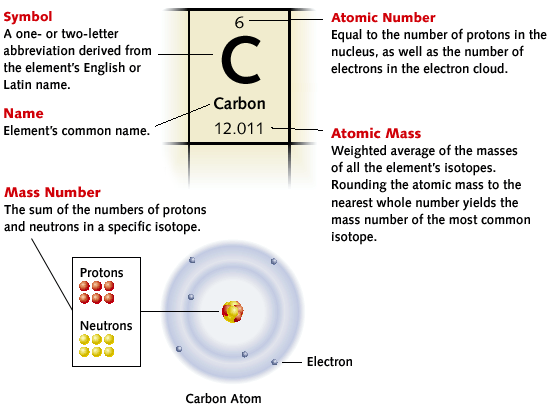
1.2 Atomic Structure
Homework for section 1.2
10GR HomeworkHW 1.2a: Workbook Pages 8 (#2), 9, 11
HW 1.2b: Workbook Pages 11, 12-13 (Q 3,4,8 only) and 19 |
10BE HomeworkHW 1.2a: Workbook Pages 8 (#2 only) and 9
HW 1.2b: Workbook Pages 11, 12-13 (Q 3,4,8 only) and 19 |
|
By the end of this section you should:
|
|
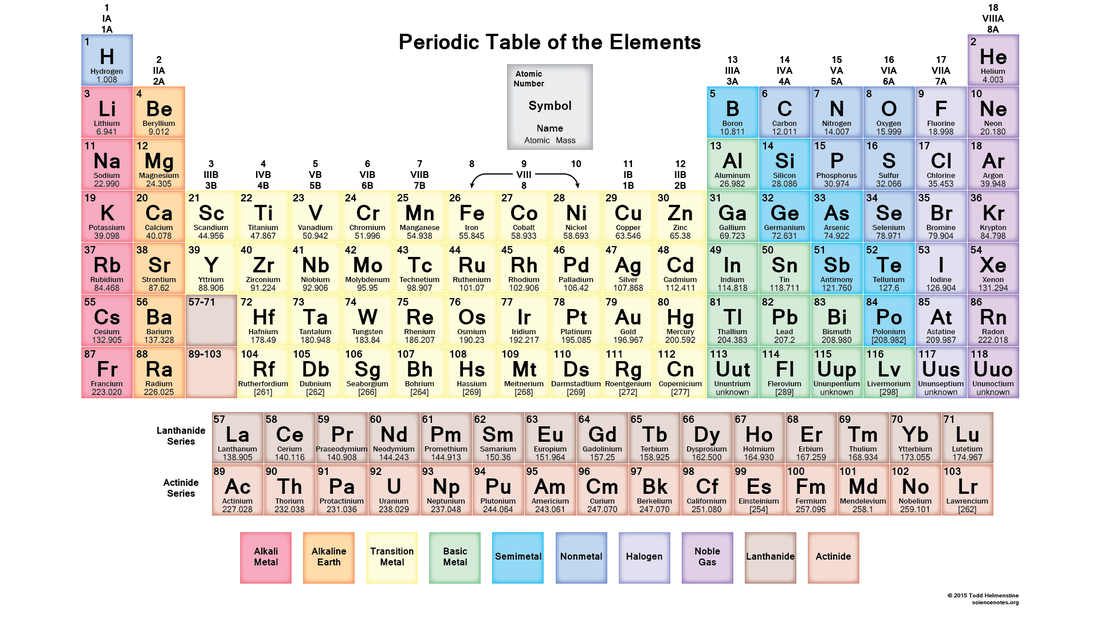
1.3 Periodic Table
Homework for section 1.3
10GR Homework |
10BE Homework |
|
By the end of this section you should know:
Key points about the Periodic Table
|
|
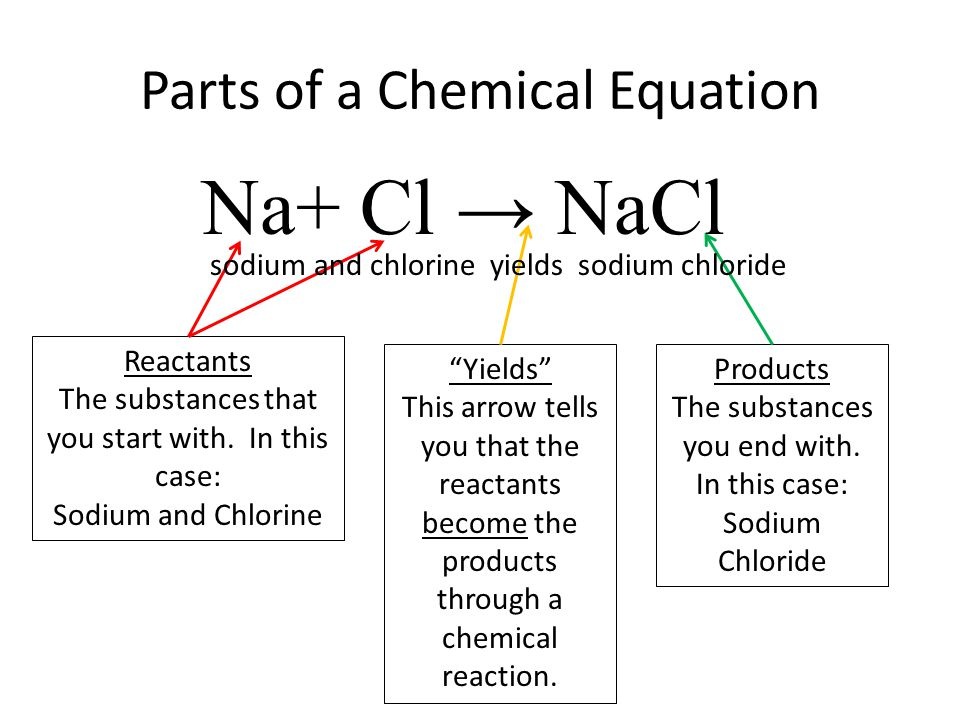
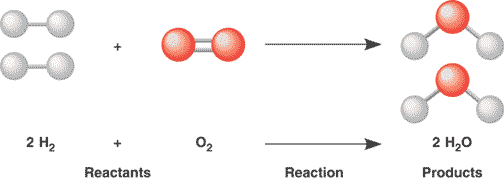
1.4 Reaction of Metals
Homework for section 1.4
10GR Homework |
10BE Homework |
|
By the end of this section you should know:
Balancing a Chemical Equation |
|
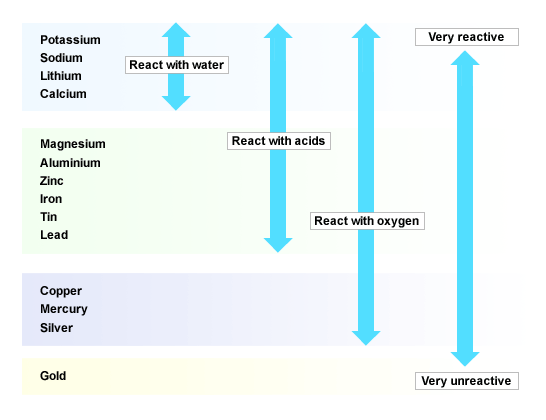
1.5 Reactivity of Metals
Homework for section 1.5
10GR Homework |
10BE Homework |
Suggestions for reviewing for the exam
Make sure you have completed the assigned workbook pages: 8,9,11,14,19,24,42
Also look at some review and extra practice pages: 16,22,23,25,29-30
Read the SLO's and Vocab lists at the top of this page and make sure you can answer all the questions and define all the vocabulary
You can also watch the videos on this page to help your study
Also look at some review and extra practice pages: 16,22,23,25,29-30
Read the SLO's and Vocab lists at the top of this page and make sure you can answer all the questions and define all the vocabulary
You can also watch the videos on this page to help your study